 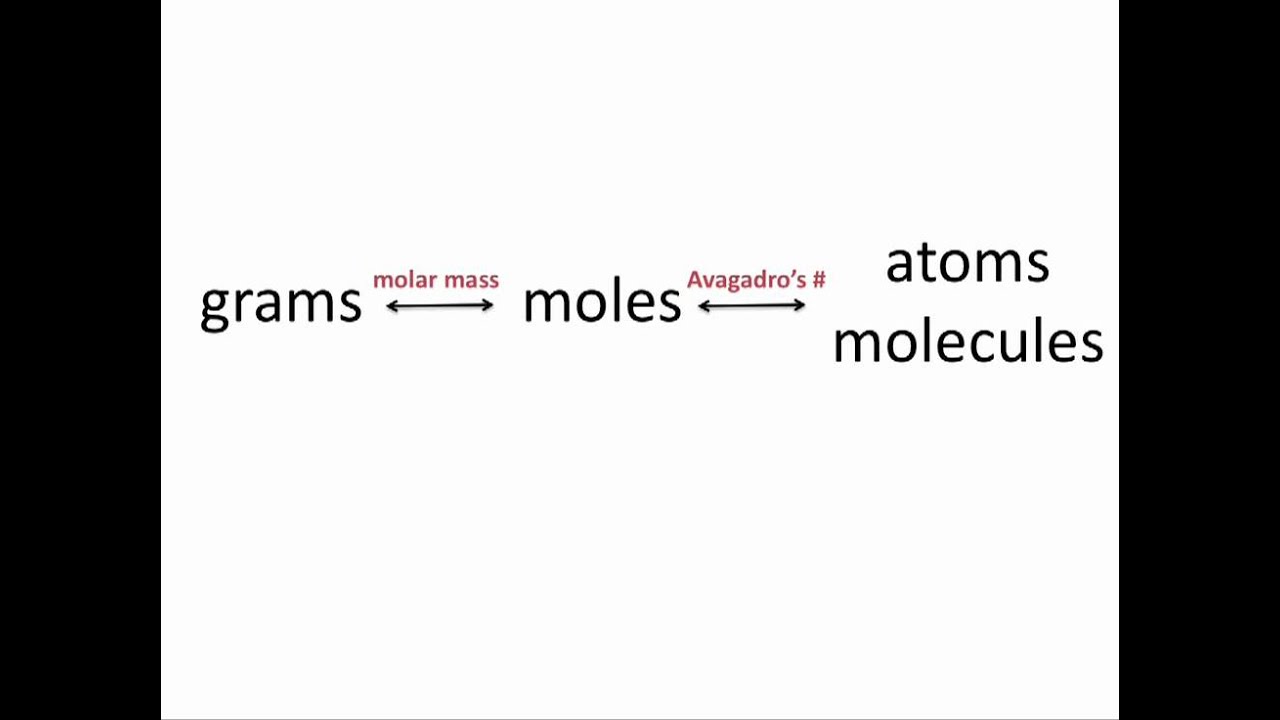
Any substance is made up of a large number of atoms, molecules, or other components, the mole is a useful measure to utilise in this case. What is a Mole?Ī mole is 6.023*1023 of any chemical unit, including atoms, molecules, ions, and others. This is the reason why we convert given grams of substance into moles and this gram to moles calculator tool by Testbook aids that purpose. The earlier approach (mole approach) helps us with the same. The latter approach (using mass) provides no information regarding the proportion of each species' quantities that react to produce the given amount of liquid water. With the 1st approach, here we have 2 moles of hydrogen reacting with 1 mole of oxygen to produce 2 moles of water.Īs per the 2nd approach, if we consider mass, we have 4g of Hydrogen gas in reaction with 32g of Oxygen gas to result in 36g of liquid water. To analyze the above equation, we have two approaches - using mole and unison mass. Let’s go through the example given below for a better understanding of mass and moles in a chemical reaction. The number of atoms, molecules, or ions that are actually reacting to produce a specific number of atoms, molecules, or ions is not indicated by the use of mass alone. 
It would be convenient to know the precise ratio of the amounts of each species when examining reactants and products in a chemical process. Why do we use the Grams to Moles Calculator? Step 3: In the output field, the grams to moles conversion will be shown. Step 2: To obtain the conversion value, click the "Solve" button now. Step 1: Fill in the input field with the grams and formula weight. Testbook’s grams to moles converter should be used as follows: You can convert grams to moles calculator with the steps mentioned below. Along with the free online calculator tool, the article also focuses on manual methods to convert grams into moles, solved examples and some brainstorming FAQs. We got you! Testbook provides you with this facility with the help of grams to moles calculator which is super quick and easy to use. Converting grams into moles can be tricky. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
